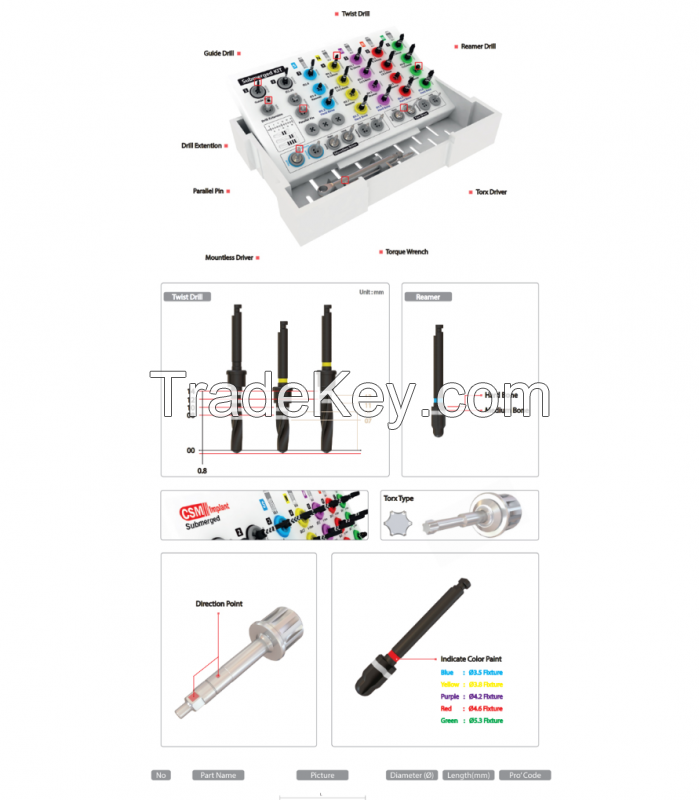
Submerged implant kit is a comprehensive set of implants which are
necessary for dental implant surgical procedure.
Our submerged implant kit consists of basic drill, final drill,
reamer and prosthetic. Basic drill included in submerged implant
kit determines initial entry point on the cortical bone. Final
drill has an excellent drilling capability provided by optimal
design of the tips. Reamer creates spaces for upper part of
fixtures for easy implantation, and prosthetic is used when
implanting fixtures. There are many models of implant devices.
Check our website for our submerged implant kit.
The CSM Submerged Implant System includes various one-stage xtures
and two-stage xtures made of titanium. These implants are inserted
into the upper and/or lower jawbone and served as a tooth root
replacement providing a stable foundation for restorations.
CSM Implant System Instructions for Use
Precautions
Surgical technique for endosseous dental xture implant requires
special and complex procedures. Formal training for xture placement
is recommended. Important : Determine local anatomy and suitability
of the available bone for xture placement. Thorough screening of
prospective xture must be performed. Panoramic and periapical
radiograph for visual inspection are essential to determine
anatomical landmarks, occlusal condition, periodontal status and
adequacy of bone. Lateral cephalometric radiographs, computerized
axial tomograghy and tomogram could be instructive. Appropriate
radiography, direct palpation and visual inspection of the xture
site are necessary for planning or treatment prior to use
xtures.
Directions for use
The surgical procedure should be done under aseptic condition with
specially designed sterile surgical instruments. The sterilized
implants have to be delivered precisely from package to preparation
site. An electrical surgical drilling system with internal or
external irrigation is recommended. Prescribed drilling sequences
(Guide drill Pre-nal drill - Final drill) or combination of
surgical tools with 70Ncm torque power should be followed to make a
hole in the jawbone and to place a xture. In case of Hard Bone(D1)
please use CSM D1 Drill or the drill of next diameter. Parallel Pin
is used for measuring the direction and the depth of the hole.
Fixture in the ampul should be placed up by using Mountless Driver
and be planted into the bone. Handpiece or Ratchet Wrench is usable
to plant. Insertion depth of CSM xture shall be 0.2mm below than
the bone level. When the xture is fully seated, carefully remove
Mountless Driver and place Cover Screw or Healing Abutment on the
xture. Then close tissue ap and suture it. The healing period will
be 45~90days for lower jaw, 90~180days for upper jaw. (Don't lost
balance in drilling work during operation)
Usage and Storage
• This product is disposable.
• It should not be reused.
• All the product package should be stored at low humidity and room
temperature condition
Caution
• Surgical Complications : Implant procedure has risks such as
localized swelling, dehiscence, temporary pain, edema, hematoma or
bleeding. Numbness of the lower lip and jaw after mandible surgery
and tissue beside the nose after maxilla surgery is a possible
side-eect. It is a temporary nature, though the numbness lasts
permanently very rarely. Gingival-mucosal (gum tissue) ulceration,
tissue reaction, or infection may occur but it generally responds
to local care.
• After the procedure of implant treatment could be found
swollenness of a specic part, rupture, temporary palpate
sensitiveness, an edema, hematoma, bleeding.
• Insensibility of lower jaw and lip, some side eects relating
around the nose from maxillary sinus treatment may occur that is
mostly temporary but rarely permanent paralysis could
appear.
• Contraindications : Fixture should not be placed in case where
the remaining jaw bone is too diminished to provide adequate width
and height to surround the implant. Lack of osseointegration or
subsequent implant failure may occur in cases insucient bone
available, poor bone quality, poor oral hygiene, heavy smoking, or
under medical conditions such as blood disorders, infected HIV
virus or uncontrolled diabetes.
• Warnings (Wornings for possible side eects from the usage of
medical devices) : As xture surgery and dental restoration involve
complex surgical procedures, for safe and eective use of xtures,
specialized training is strongly suggested. Improper patient
selection and technique can cause the xture failure and/or loss of
supporting bone. Wobble of xture, bone loss, or chronic infection
may cause xture failure. If the xture becomes contaminated by the
patients body uids in any way, the xture cannot be used in other
patient. Do not splint or bridge with natural tooth. Excessive
force on the xture will cause metal fatigue and can cause xture
fracture. Single stand or by xtures are recommended.
• Interaction : Dental xtures are designed only for its use and
must not be altered and converted in any way. The use of
electro-surgical or laser surgical instruments around metallic
xtures and their abutments is not recommended due to the risk of
electric shock and/or heat burn.
• Usage for the aged or a pregnant woman : The aged, a pregnant
woman, growth phase, an obese patient or a patient who had other
case history shall be considered to perform an operation.
• Warnings for application : A surgeon shall be followed
instruction of the procedure for high success rate of implant
surgery. Advert to not damage on nerves, diagnose concavity on
lingual side of the submaxillary bone for preventing perforation of
the submaxillary bone. Do not put over torque for preventing bone
crack. • Caution of safety accidents : Take any possible stus away
as combustibility materials or unsterilization devices from a
patient and a surgeon.